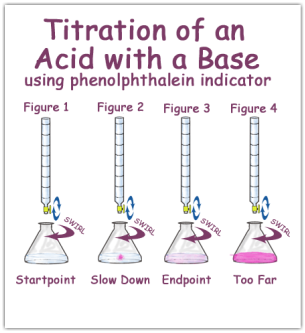
A titration is a method of analysis that will allow you to determine the precise endpoint of a reaction and therefore the PRECISE quantity of reactant in the titration flask. A burette is used to deliver the second reactant to the flask and a suitable indicator is used to INDICATE the endpoint of the reaction.
We react the ACID with an alkali using a suitable indicator.
Things we NEED to know:
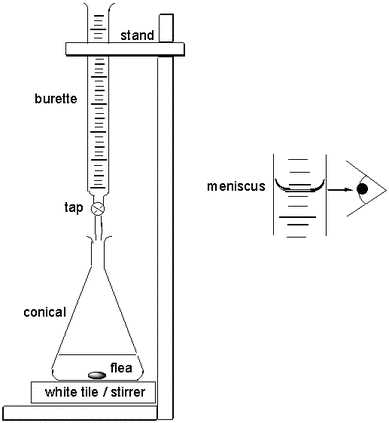
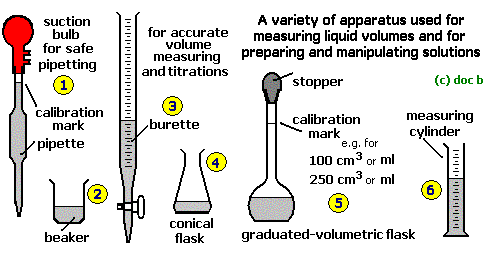
Apparatus:
We react the ACID with an alkali using a suitable indicator.
Things we NEED to know:
- [acid]
- the REACTION balanced equation between the acid and alkali
Apparatus:
Steps of a titration:
This website link below shows you a detailed step by step version with appropriate images to help you understand what is involved in a titration:
- Put GOGGLES (eye protection) on. Use equipment with CARE and assume that all solutions are TOXIC and CORROSIVE.
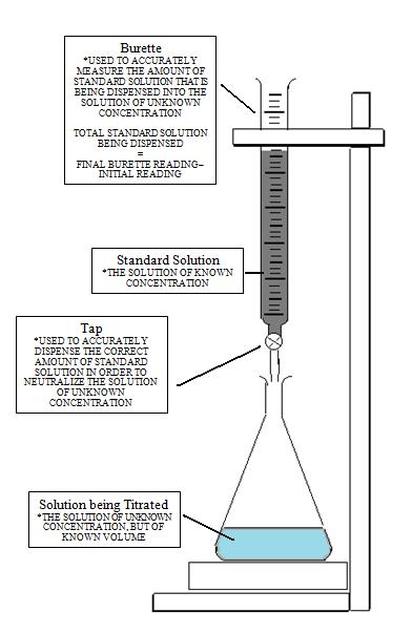
- Close the STOPCOCK (where it stops the solution from coming out of the BURETTE).
- When you are happy that the BURETTE is below EYE LEVEL, use a FUNNEL to RINSE the BURETTE with the substance you are trying to find the [ ] of, i.e ACID (e.g sulfuric acid) as its [ ] is UNKNOWN.
- FILL the BURETTE to an appropriate level. Make a note of the INITIAL burette reading in a table.
- Use a PIPETTE FILLER to RINSE the pipette with the ALKALI (NaOH solution). ACCURATELY measure the VOLUME of the ALKALI (of known [ ], e.g 25.0 cm3 of NaOH solution) and TRANSFER into a (250 cm3) CONICAL FLASK using the CALIBRATED PIPETTE and PIPETTE FILLER.
- Add 3 or 4 drops of suitable INDICATOR (e.g phenolphthalein) to the CONICAL FLASK (with the ALKALI in it).
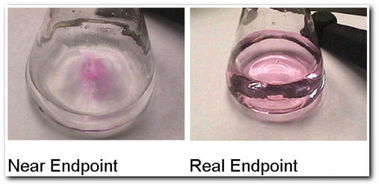
- Run in ACID from the BURETTE into the CONICAL FLASK (contining the ALKALI) until the COLOUR of the solution in the CONICAL FLASK changes (from colourless to light pink). This shows that the SOLUTION in the conical flask is now NEUTRAL. Note: you will know when you have NEARLY reached NEUTRALISATION as the when you add more ACID (from the BURETTE), it will add to the colourless solution as a FLASH OF PINK that will go away with stirring.
- Record your final burette reading in your table.
- Rinse the conical flask with distilled or de-ionised water.
- REPEAT this procedure until you obtain 2 TITRES (VOLUME of ACID used at NEUTRALISATION) that are within 0.10 cm3 of each other.
- Calculate and record the AVERAGE titre from the CONSISTENT TITRES.
This website link below shows you a detailed step by step version with appropriate images to help you understand what is involved in a titration: