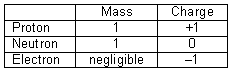
SUB-ATOMIC PARTICLES
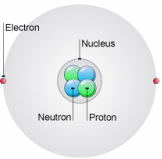
You should already know from GCSE that atoms are made of these fundamental particles:
You should already know from GCSE that atoms are made of these fundamental particles:
- proton* *= KNOWN AS NUCLEONS, form the nucleus
- neutron*
- e- = surround nucleus, in levels known as orbitals
The number of energy levels/shells of an atom corresponds to the period the atom is in. For e.g Na is in the 3rd Period, so has 3 shells.
Each sub-level of the atoms contain ORBITALS, which can each hold 2 e-.
Below shows the key information you need to know:
Energy Level Sub-levels Max no of e- in this level
n=1 2 1s
n=2 8 2s, 2p
n=3 18 3s, 3p, 3d
n=4 32 4s, 4p, 4d, 4f
Each sub-level of the atoms contain ORBITALS, which can each hold 2 e-.
Below shows the key information you need to know:
Energy Level Sub-levels Max no of e- in this level
n=1 2 1s
n=2 8 2s, 2p
n=3 18 3s, 3p, 3d
n=4 32 4s, 4p, 4d, 4f
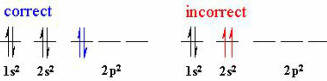
Rules for e- arrangement:
- Auf bau principle: orbitals with the lowest E are filled first, e.g 1s will be filled before 2s.
- Pauli principle: e- have a property 'SPIN', e.g if 2 e- occupy an orbital their 'spins' will be opposite (otherwise they would repel).
- Note: draw e- as arrows.
- Hund's rule: if multiple orbitals of the same E are available, they are occupied by single e- where possible (e- don't share orbital with other e- unless have to).
- highest E e- are lost 1st. Cr & Cu are exceptions to the rule as it is beneficial for them to donate e- to the higher E orbital as there is less repulsion.
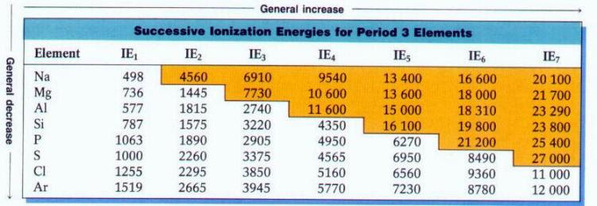
Ionisation Energy (I.E)
1st I.E= the E required to remove 1 mole of e- from 1 mole of atoms in the gaseous state!
1st I.E= the E required to remove 1 mole of e- from 1 mole of atoms in the gaseous state!
Mass number, A:
A- TOTAL number of protons and neutrons / NUCLEONS (in nucleus of
atom)
Atomic/Proton number, Z:
A- number of PROTONS and no of e- (if atom has no overall charge)
Relative atomic mass:
A- Average mass of 1 atom (of an element)/ one twelfth the mass atom of an atom of 12C
Isotope:
A- ATOM with the SAME no of PROTONS, DIFFERENT no of NEUTRONS
A- TOTAL number of protons and neutrons / NUCLEONS (in nucleus of
atom)
Atomic/Proton number, Z:
A- number of PROTONS and no of e- (if atom has no overall charge)
Relative atomic mass:
A- Average mass of 1 atom (of an element)/ one twelfth the mass atom of an atom of 12C
Isotope:
A- ATOM with the SAME no of PROTONS, DIFFERENT no of NEUTRONS
State why isotopes of an element have the same chemical properties:
A- SAME number of e- (in outer shell) and e- are RESPONSIBLE for CHEMICAL PROPERTIES
A- SAME number of e- (in outer shell) and e- are RESPONSIBLE for CHEMICAL PROPERTIES