|
The mass spectrometer is used to determine the relative Ar of atoms and Mr of molecules accurately. Relative Mr's are measured relative to Carbon as it is the only element in the periodic table to have a mass of EXACTLY 12, a WHOLE number.
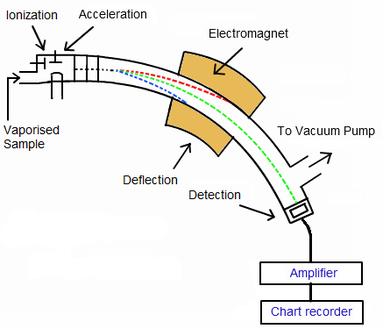
The mass spectrometer determines the mass of separate atoms. This is useful for forensic scientists to identify substances, like illegal substances. The mass spectrometer is kept under a high vacuum to make sure the ions do not react with air molecules and ensure they reach the detector. Remember: Victor Is A Daft Duck (VIADD) |
VAPOURISATION:
- The sample must be in the GASeous state.
- If it is not a (g) already it must be vapourised by HEATING.
- A beam of HIGH E e-'s are fired from an e- gun to form POSITIVE IONS.
- Usually only 1 e- is knocked out of each atom, but on occasion 2 or more e- may be knocked out, which affects the m/z value.
- As we only need one e- to be knocked out, no more than the minimum energy is used to ionise samples.
- This step is needed so that acceleration, deflection and detection can occur.
- The positive ions are ATTRACTED towards NEGATIVELY charged plates and accelerated at HIGH SPEED.
- Lighter ions move faster.
- A MAGNETIC FIELD deflects the ions in varying amounts, depending on their M/Z values.
- m/z=mass of atom to charge of atom ratio.
- Ions with the lowest m/z value (lighter ions) are DEFLECTED MORE than ions with a higher m/z value (heavier ions)
- Note: an ion with a +2 charge will be DEFLECTED TWICE as much as an atom of +1 charge of the same mass.
- The deflection also depends on the STRENGTH of the magnetic field. A STRONGER magnetic field will give a GREATER DEFLECTION.
|
DETECTION:
|