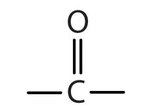
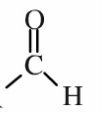
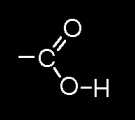
- Functional grp= OH (hydroxyl)
- Soluble in H2O- due to H bonding with OH grp. Alcohols are more soluble the shorter the chain length, so less soluble the longer the chain length-due to it only having 1 OH grp on it.
Nomenclature of Alcohols:
Suffix: Classification:
Suffix: Classification:
- generally: ol
- has <2 C atoms, locant required e.g propan-1-ol
- (alternate prefix= 'hydroxyl' e.g 1-hydroxylpropane)
- has more than 1 OH grp e.g propane-1,2,3-triol
- has double bond e.g but-3-en-1-ol (3=where C=C, 1= where OH grp)
- just note: OH takes priority when naming, so if on an end C there is an OH functional grp and a Fluro functional grp on the other end C, then you take the OH's C to be C1.
Classification (due to structure):
(Basically how many C's are attached to the (special) C with the OH on it
- 1*- has 1 C attached to the (special) C with an OH on it
- 2*- has 2 C's attached to the (special) C with an OH on it
- 3*- has 3 C's attached to the (special) C with an OH on it
(Basically how many C's are attached to the (special) C with the OH on it
Production of Ethanol:
|
1) FERMENTATION of Sugar:
C6H12O6 --> CH3CH2OH + 2CO2 Conditions:
Note: you should be aware of the C neutral argument-that in equations the C taken in is= to the C released into the atmosphere, BUT not realistic as does not take into consideration the C released from vehicles to transport the ethanol.
|
2) HYDRATION
C2H4 + H2O(g) <--> C2H5OH Conditions:
|
Note: the strengths for one are the weaknesses for the other and vice versa.
REACTIONS OF ALCOHOLS:
Combustion:
X + 0xygen --> CO2 + water
Oxydation:
Conditions:
Combustion:
X + 0xygen --> CO2 + water
Oxydation:
- 1* and 2* alcohols readily undergo these reactions as it involves breaking C-H bonds
- 3* alcohols not easily oxidised as involves breaking C-C bonds (stronger than C-H bonds)
Conditions:
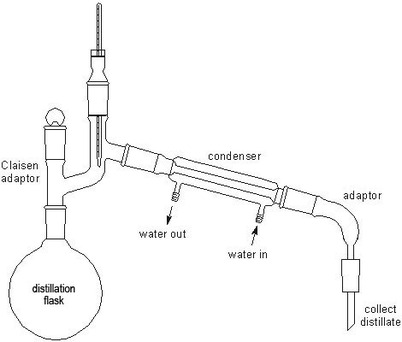
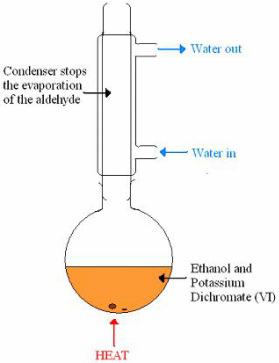
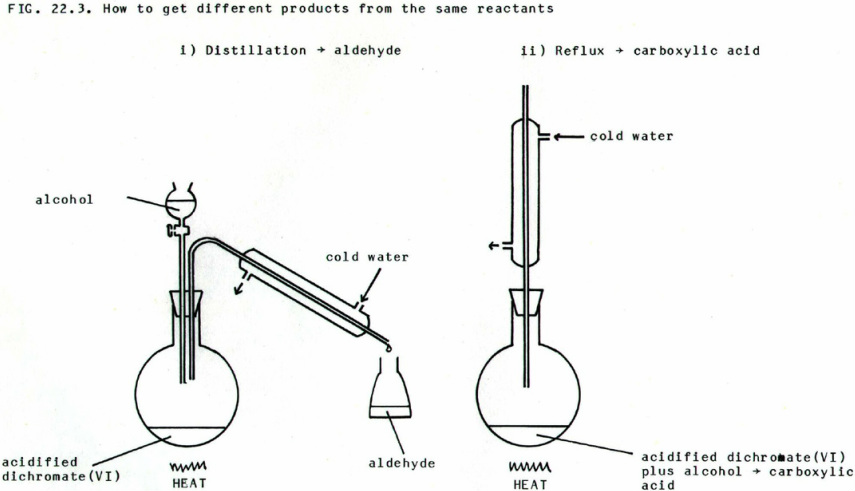
- oxidising agent= H+/K2Cr2O7 (VI) (acidified K dichromate VI)
Observation: Orange --> Green
|
Tests:
1) Tollens' Reagent= Tollens' reagent (colourless) Observation=
|
2) Fahlings' Reagent= Fahlings' reagent (blue) Observation=
|
Note: as there is no change with the Ketones in either Test, remember 'There's no changing Keith Lemon'