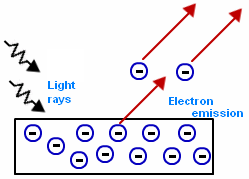
This is where e- are emitted from a metal surface as the electromagnetic radiation (containing photons) directed at the metal is above a certain f (fo).
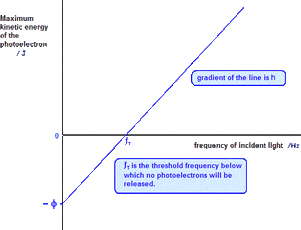
There are some equations to know for this. One is: Ek=hf-ϕ.
This is the same as the mathematical equation: y=mx+c. It makes sense that the work function is a minus (-) as in this graph below you can see that the work function is below zero: here to edit.
This is the same as the mathematical equation: y=mx+c. It makes sense that the work function is a minus (-) as in this graph below you can see that the work function is below zero: here to edit.
You should be aware that:
- 1 photon is absorbed by 1 e-.
- if f>fo and you increase f, then according to E=hf, hf will increase, so Ek max will increase.
- if f>fo and you increase the intensity of the photons fired? at the metal surface, then more e- will be released BUT Ek(max) will stay the same as your not changing the f in E=hf, so E cannot change. Note: intensity ∝ to number of e- emitted
- photons have fixed E=hf.
- the ϕ has a different value depending on the metal, but for each metal it stays constant in the equation.
- all e- will have E=hf when it has absorbed a photon, it will use and thus lose E to move to the surface of the metal and escape.
- e- from within the metal will require E to get to the surface of the metal. Depending on the E it has left, will determine whether it is emitted from the metal surface. e- at the surface of the metal leave the metal surface with E=hf of the photon as it has not lost any E to move, so it is emitted with Ek(max). This is why we get e- emitted with a range of Ek up to a maximum value (Ek(max)).
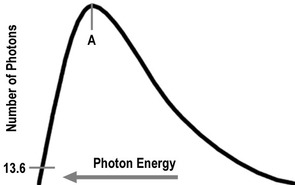
- Below shows the distribution of e- and of what E. Note: the curve would never reach zero as in theory particles have ∞ E.